
Draw the Lewis structure & Molecular geometry for HCN: explain the configuration in the molecule of valence electrons around the atoms
A chemical compound with the formula HCN is hydrogen cyanide. It is also called prussic acid. It is a liquid that is colorless, highly toxic, and flammable in nature. HCN is a particularly valuable predecessor of numerous chemical compounds ranging from polymers to pharmaceuticals and is processed on an industrial scale.
HCN has a slight bitter almond-like smell due to a recessive genetic mutation that certain individuals are unable to identify. The explosive agent has been used for rodenticide inhalation and human venom, as well as for the slaughter of whales. Cyanide ions interact with respiratory enzymes containing iron as well.
In a variety of plants, this HCN chemical is present in small amounts, particularly in stone fruits such as cherries, as well as in the roots of cassava. Prolonged exposure to a small quantity of cyanide can contribute to chronic health problems; in individuals whose diet includes large concentrations of cassava.
Since it prevents cellular oxidative processes, hydrogen cyanide is extremely toxic. Without severe effects, an adult person can tolerate 50 to 60 parts of hydrogen cyanide per million parts of air for an hour, but exposure to concentrations of 200 to 500 parts per million of air for 30 minutes is typically lethal.
Sources and uses of HCN
- In certain foods and in some plants, cyanide is released by natural compounds. Popular fruit seeds, such as apricots and apples, can contain large quantities of chemicals that are metabolized into cyanide.
- Cyanide is used in the production of paper, photography, plastics, etc. In metallurgy, cyanide salts are used for electroplating, washing metals, and scraping gold from the ore. It is also used in homes to eliminate rodents.
- For certain salts, hydrogen cyanide is an ideal solvent, but it is not commonly used as a solvent owing to its toxicity.
- They may also be extracted from plants in limited concentrations, where they exist in association with sugars.
- Chemicals found in artificial nails remover, if mistakenly ingested, can cause the production of hydrogen cyanide in the body.
- Cyanide is found in tobacco smoke and industrial materials such as plastics are combustion ingredients. Products of combustion are compounds that are given off as objects ignite.
Lewis Structure of HCN
The Lewis HCN structure helps to explain the configuration in the molecule of valence electrons around the atoms. It also helps to consider the bonds formed in the molecule and the electrons that do not take part in the forming of either bond.
Firstly to make the dot structure you need to decide the center atom and then position the leftover atoms in the structure of HCN. Carbon takes the middle position as it is the least electronegative. Then, you need to position the atoms of hydrogen and nitrogen on both ends of the carbon atom.
H C N
If the atoms have been arranged, start putting the dots around individual atoms representing the valence electrons. Thus, carbon will have four electrons, hydrogen will have one, and nitrogen will have five electrons inside the atom.
H: C::: N:
Now, by forming a single hydrogen bond and a triple nitrogen bond, Carbon has a full octet. Similarly, nitrogen has a full octet and it only requires three electrons to complete the octet it gets by exchanging the electrons with carbon. In its exterior valence shell, hydrogen has two electrons, and the other two electrons are non-bonding electrons.
Working of Cyanide
- The degree of cyanide toxicity depends on the quantity and duration of cyanide to which an individual is exposed.
- Cyanide gas breathing does the most pain, but it may also be poisonous to swallow it.
- Especially, in confined areas, it is risky.
- In open fields, cyanide gas evaporates instantly, making it less dangerous outdoors.
- Prevents oxygen from using the body cells owing to which the cells die.
Molecular geometry of HCN
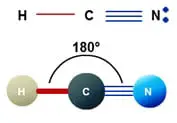
Every molecule’s molecular geometry helps to explain its dimensional structure and its form. HCN has the geometry of AX2 molecule, where the central atom is A, and the number of atoms bound to the central atom is X. It fits the molecular geometry of AX2 since carbon is bonded to two atoms. And its linear molecular geometry, as per the principle of VSEPR. Therefore, HCN has a linear molecular geometry.
How do you subject yourself to cyanide?
- Consuming food, water etc. that has HCN in it could expose you to cyanide.
- As a product of both natural causes and human practices, cyanide enters water, soil, or air and is also present as gaseous HCN in the air.
- For those who do not work in cyanide-related sectors, smoking tobacco is potentially one of the main causes of cyanide toxicity.
Why is HCN polar?
Electronegativity of Hydrogen Cyanide:
- Hydrogen – 2.1
- Carbon – 2.5
- Nitrogen – 3
Hydrogen will never assume a center position, as it is the least electronegative. And because of the difference in electronegativity between hydrogen and carbon, the charge representing the vector would be taken from hydrogen to carbon. And in the same manner, the vector would shift from carbon to nitrogen due to the reason that nitrogen is more electronegative.
Despite only a minor difference in the electronegativity of Carbon and Nitrogen, since Nitrogen would attempt to draw the electrons to itself, it is called a slightly polar bond. And the vector shifts from nitrogen towards hydrogen, because hydrogen has a positive charge and nitrogen, has a negative charge. And if any molecule has an electronegativity difference from the dipole’s side it is said to be a polar molecule. Therefore, HCN is a polar molecule.
Conclusion
In order to sum up a few points in this article, we can say:
- One single bond with the Hydrogen atom is formed by carbon and a triple bond with the Nitrogen atom is formed.
- Cyanide is usually found in natural compounds such in the seeds of the fruits.
- It is enclosed and has a linear structure under the AX2 molecular geometry.
- HCN is a molecule that is polar in nature.


